MAIN TOPIC: EVOLUTION OF PHARMACOVIGILANCE: INTEGRATING NEW DATA SOURCES, POPULATION STUDIES AND PREDICTIVE TECHNOLOGIES
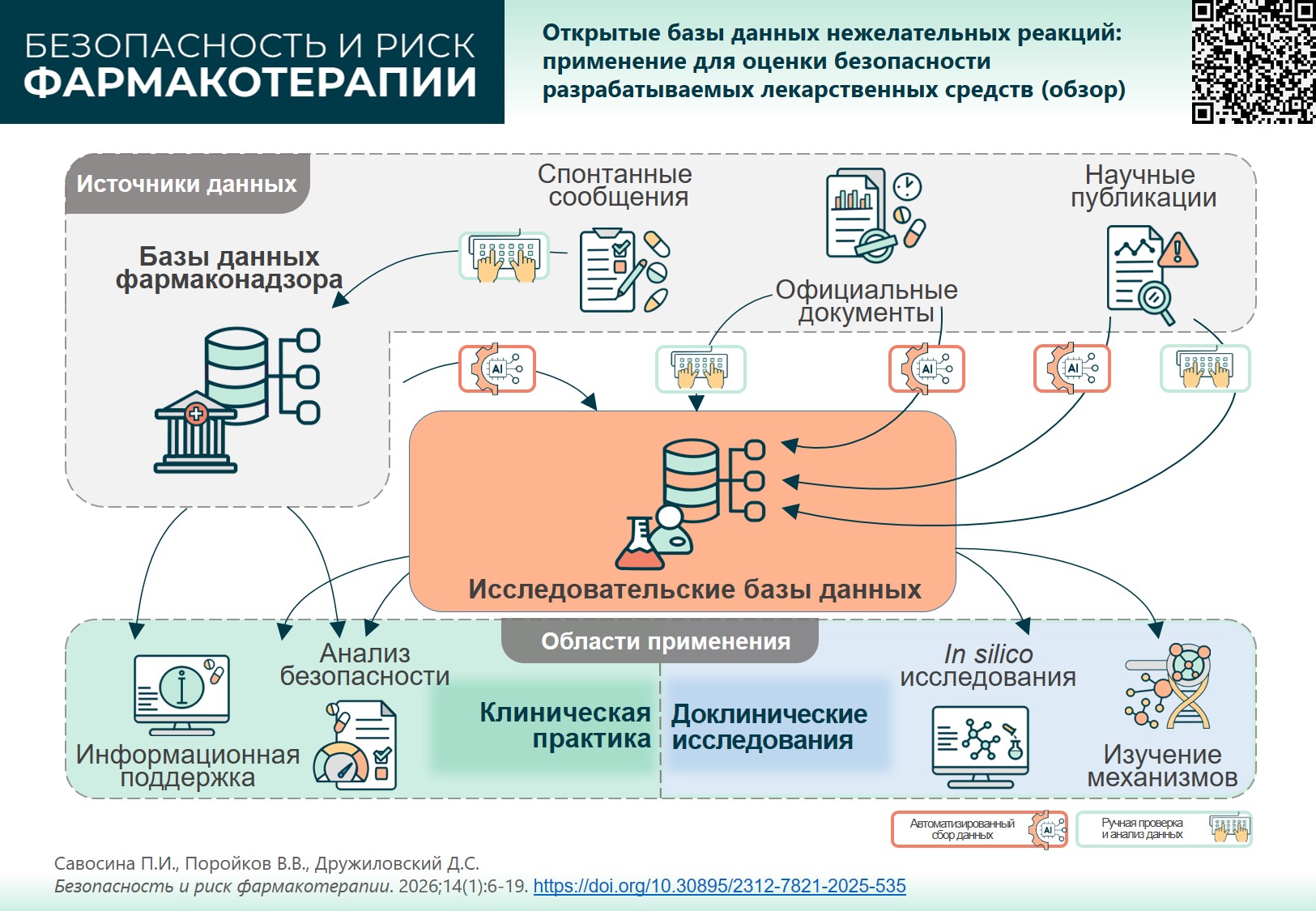
INTRODUCTION. Evaluating safety data on already registered drugs is an important stage in medicine development. Databases (DBs) of adverse drug reactions (ADRs) have been created and maintained by regulatory authorities and individual research groups. The data aggregated in such DBs can help create reference and training datasets for experimental and in silico studies. A preliminary assessment and systematisation of information from these databases can facilitate the selection of DBs relevant to a specific study.
AIM. This study aimed to assess applicability of public ADR databases for safety studies of medicinal products.
DISCUSSION. Eleven public DBs that provide information on ADRs of approved drugs were studied: FAERS, DAEN, MedEffect Canada, EudraVigilance, VigiBase, SIDER, MetaADEDB, ADReCS-Target, T-ARDIS, OnSIDES, and WWAD. The differences between these databases are primarily due to the variety of sources they use: spontaneous reports (FAERS, DAEN, MEDEFFECT, EudraVigilance, VigiBase), patient information leaflets and other official documents (SIDER, OnSIDES, WWAD), scientific publications (ADReCS-Target), and other open-access web resources (MetaADEDB, T-ARDIS). All the reviewed databases can be used for informational support and analysis of drug safety profiles. SIDER, MetaADEDB, ADReCS-Target, OnSIDES, and WWAD are useful in preclinical studies, particularly while developing training sets for in silico methods. Hypotheses for possible ADR mechanisms and search for new drug repurposing vectors can be arranged using ADReCS-Target, WWAD, and T-ARDIS, since these DBs provide additional data on active pharmaceutical substances or target molecules. Using computer-aided methods without thorough hands-on search in the DBs such as ADReCS-Target, T-ARDIS, OnSIDES, and MetaADEDB, limits their applicability in tasks requiring accurately analysed information.
CONCLUSIONS. The DBs reviewed can serve as a valuable tool for addressing a wide range of biomedical issues. To select a DB relevant for a specific study, it is important to consider the underlying principles, since varying sources and annotation methods can affect the reliability of results.
INTRODUCTION. Inconsistent assessment of adverse drug reaction (ADR) remains a relevant pharmacovigilance challenge, particularly regarding insulin biosimilars widely used in the medical practice. This is especially critical for ADRs associated with the pharmacological effects of insulin (hypo- and hyperglycemia) and hypersensitivity reactions, frequently classified as serious due to a lack of adequate clinical context. This discrepancy reduces analytical value of the available data and results in consequent accumulation of inaccurate safety data on a medicinal product.
AIM. This study aimed to identify quality improvement approaches to data provided in spontaneous reports and to compare the assessment of ADR seriousness for insulin preparations (insulin aspart, insulin lispro, and insulin glargine) performed by reporters and by pharmacovigilance department of the marketing authorization holder.
MATERIALS AND METHODS. A retrospective observational study was conducted using spontaneous ADR reports submitted since the registration of insulin aspart, insulin lispro, and insulin glargine manufactured in the Russian Federation (2021–2024, GEROPHARM LLC). Valid cases were coded using MedDRA v.25.1 and then analyzed. Seriousness criteria were defined according to ICH E2A Clinical Safety Data Management: Definitions and Standards for Expedited Reporting and the EAEU Good Pharmacovigilance Practice. Descriptive statistics and comparative analysis of MedDRA Preferred Terms were applied.
RESULTS. Significant discrepancies were identified: assessments by reporters and marketing authorization holder matched only in 41.3% of cases, while 58.7% demonstrated inconsistency. In 99.6% of the latter cases, reporters classified ADRs as serious, while marketing authorization holder assigned them as non-serious. The most disputable criteria selected by reporters were Important Medical Events (71.1% of all discrepancies) and Life-Threatening Events (25.0%). Disagreements predominantly involved ADRs related to pharmacological effects of insulins and hypersensitivity reactions. More than half of the spontaneous reports contained insufficient information, limiting the accuracy of medical assessment.
CONCLUSIONS. The substantial differences in seriousness assessment may be associated with insufficient quality and completeness of the submitted data, frequent use of subjective criteria (such as Life-Threatening and Important Medical Event), and persistent skepticism about biosimilar insulin products. Improving pharmacovigilance accuracy requires educational initiatives and measures aimed at enhancing the quality of spontaneous ADR reporting.
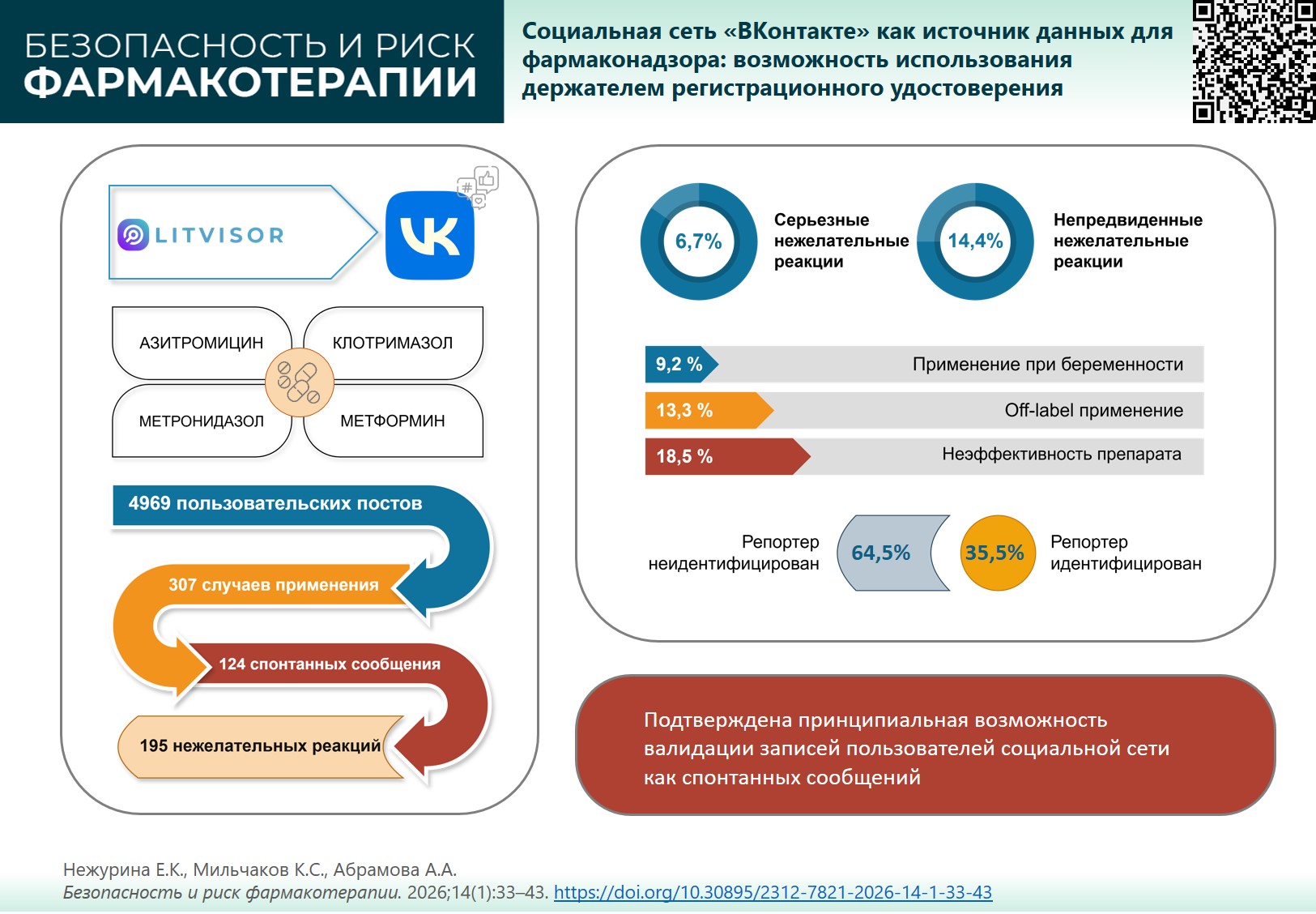
INTRODUCTION. Social media (social networks, forums, review websites, etc.) contain a lot of essential information about adverse drug reactions (ADRs). So far, no previous studies of social media were conducted in the Russian Federation.
AIM. This study aimed to evaluate the possibility of using VKontakte social network as an additional source of ADR reports exemplified by users mentioning the use of metformin, azithromycin, metronidazole, and clotrimazole.
MATERIALS AND METHODS. VKontakte social network was monitored for the period of 09.01.2023 to 03.31.2024 using LITVISOR® software to collect entries mentioning the use of metformin, azithromycin, metronidazole, and clotrimazole (International Non-proprietary Names). We analyzed the completeness of information about the reporters and the patients, as well as ADRs and special safety situations. The identified safety information was encoded using MedDRA terminology; their seriousness and listedness were assessed.
RESULTS. A monitoring of VKontakte social network resulted in 4,969 entries on the use of azithromycin, metformin, metronidazole, and clotrimazole. We identified 195 ADRs related to 15 systemic organ classes; of them, 93.3% were classified as non-serious and 6.7% as serious. 85.56% of ADRs were expected, while 14.4% were unexpected. Cases of off-label use, overdose, and use in pregnant women were identified. In 35.5% of spontaneous reports, the reporter was identified, in 89.5%, the patient’s sex was known, and in 36.3%, the patient’s age was known.
CONCLUSIONS. The findings show that the monitoring of VKontakte social network is a promising source of data on drug safety approved in the Russian Federation. The study confirms the fundamental possibility of validating the entries from the social network users as spontaneous reports.
INTRODUCTION. Pharmacovigilance ensures drug safety through continuous monitoring, yet awareness remains limited in low-resource settings. Ranitidine, a widely used antacid, was withdrawn globally in 2020 due to contamination with N-nitrosodimethylamine (NDMA), a suspected carcinogen. Despite an official withdrawal from drug circulation, ranitidine is still at risk of being used unreasonably; the conditions given make it a relevant goal to assess professional awareness of pharmaceutical employees.
AIM. This study aimed to detect pharmacovigilance challenges among Lybian pharmacies and explore the new growth vectors using awareness of ranitidine withdrawal reasons and safety issues among pharmacy employees as an example.
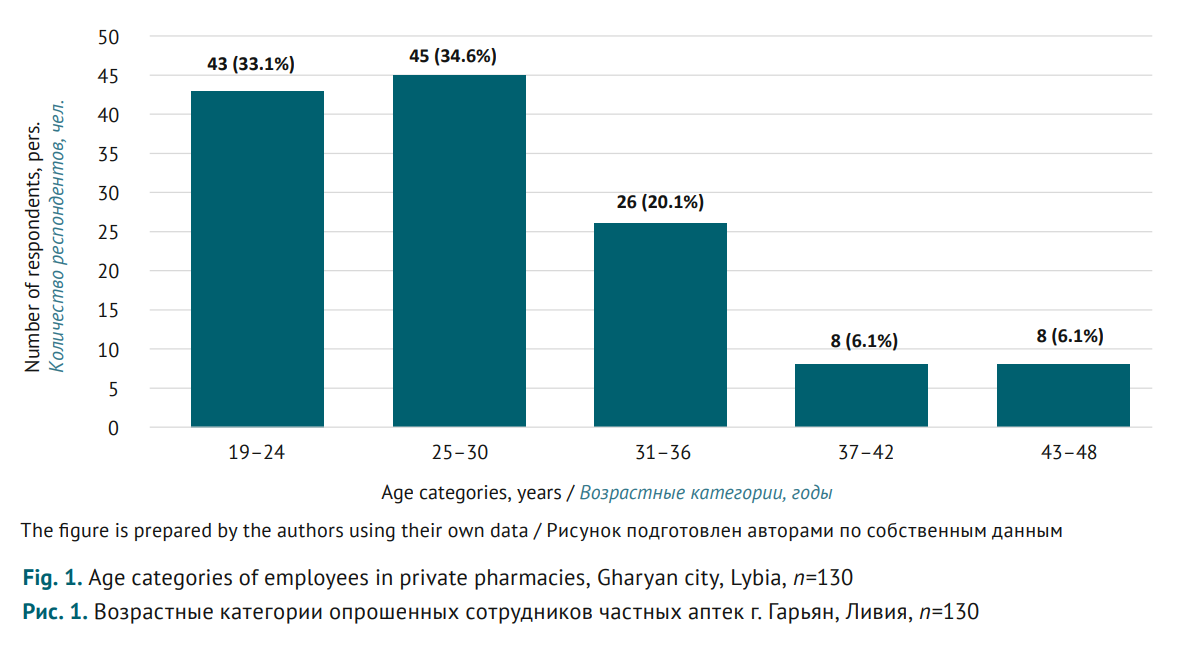
MATERIALS AND METHODS. A cross-sectional study was conducted engaging 130 pharmacy employees of Gharyan city, Libya, in January-March 2023. A structured questionnaire with three sections was used, including: 1) demo graphics (age, gender, education, and experience); 2) basic pharmacovigilance knowledge of participants (six questions with Yes/No answers); 3) awareness of ranitidine withdrawal (six questions with Yes/No answers). Data were analysed using descriptive statistics.
RESULTS. The survey showed that almost a third of participants (36.2%) were not familiar with the term “pharmacovigilance”; more than two-thirds (63.8%) did not know that Libya’s national pharmacovigilance centre existed. This contradicts the responses regarding pharmacovigilance measures: 78.5% stated they were trained in pharmacovigilance; 81.5% knew about special report forms to be filled out for any adverse drug reactions, while 16.2% previously made reports on adverse reactions. 55.4% of participants were familiar with the cases where drugs were withdrawn due to related risks. At the same time, 79.2% thought that ranitidine was still dispensed from the pharmacies; and 30.8% misclassified ranitidine, a Н2-blocker, as an antihistamine. 61.5% of participants thought carcinogenic impurities were caused by manufacturing contamination; moreover, only another 36.1% associated the impurities also with the improper storage.
CONCLUSIONS. The identified critical gaps in pharmacovigilance knowledge and ranitidine safety highlight the need for targeted educational interventions among Gharyan pharmacy employees and regulatory enforcement of drug withdrawals from the circulation.
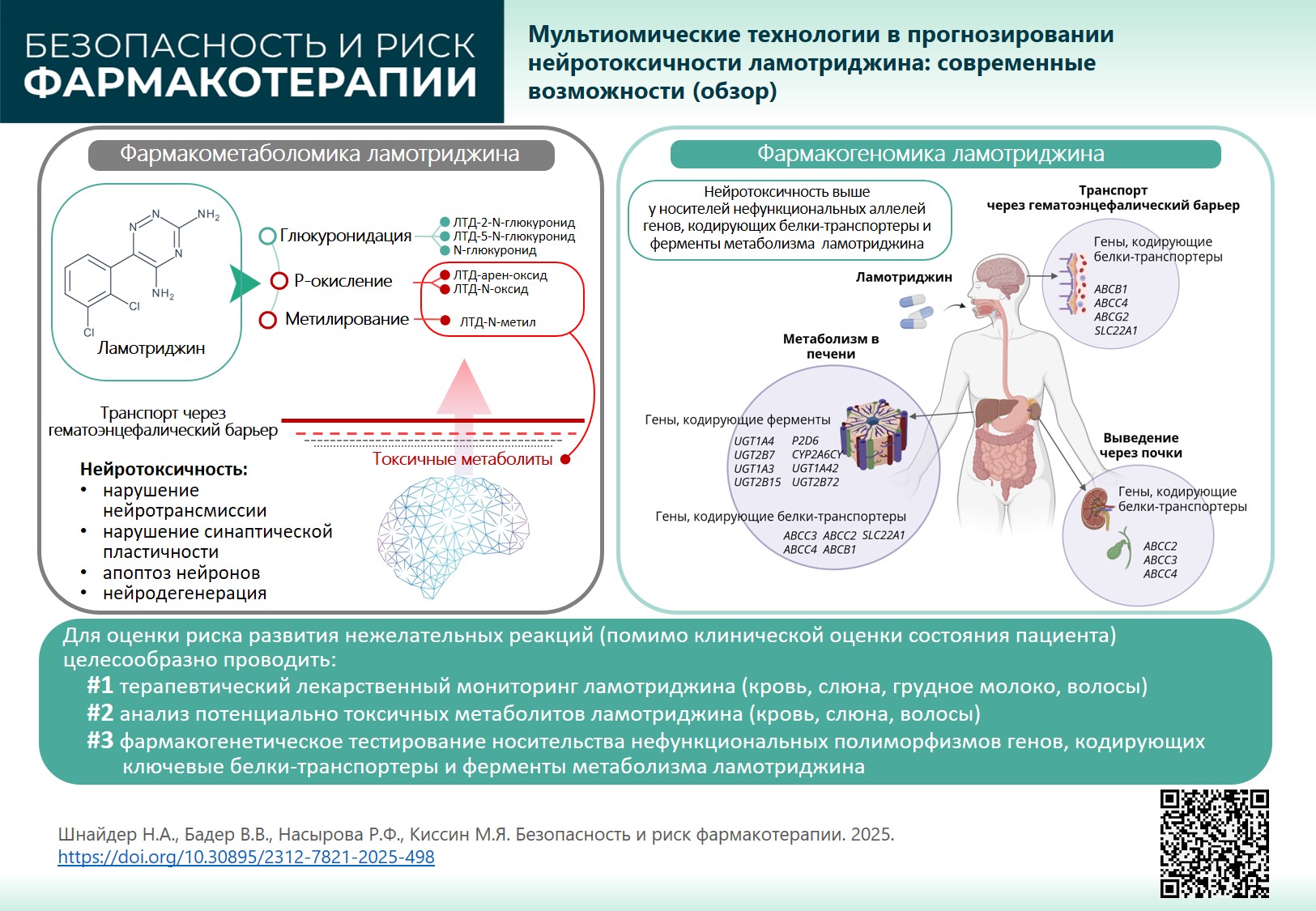
INTRODUCTION. Lamotrigine (LTG) is among the most commonly prescribed second-generation antiepileptic drugs due to its low teratogenic risk. However, lamotrigine has pronounced neurotoxic, hepatotoxic, dermatotoxic potential (for genetic and metabolic causes) and in some cases can even cause multi-organ failure. Understanding lamotrigine mechanism can help individualise therapy and increase its safety, considering pharmacodynamics and pharmacometabolomics that determine its metabolism, transport, and elimination in a particular patient.
AIM. This study aimed to develop an approach to lamotrigine therapy of epilepsy and other neurological and psychiatric diseases reducing neurotoxicity, with due regard to pharmacogenomics and pharmacometabolomics.
RESULTS. LTG is metabolised in the liver in two pathways: glucuronidation (major pathway) and P-oxidation (minor pathway). As a result, neutral and toxic (reactive) lamotrigine metabolites are produced that can circulate in blood serum for a long time, penetrate the damaged blood-brain barrier in patients with therapy-resistant seizures and have a neurotoxic effect, triggering or maintaining neurotransmission disorders, impaired synaptic plasticity, neuronal apoptosis and other neurodegeneration mechanisms. An important role in lamotrigine neurotoxicity belongs to transport proteins involved in the efflux (excretion) of reactive (toxic) metabolites from the brain into the systemic circulation, as well as from hepatocytes into the gastrointestinal tract by bile and through the kidneys with urine. Genetically determined delayed efflux through the blood-brain barrier (pharmacogenomics) increases lamotrigine neurotoxic potential.
CONCLUSION. To assess the risk of lamotrigine-induced adverse reactions, together with clinically assessing patient’s condition, it is recommended to: 1) monitor drug distribution (blood, hair, saliva, breast milk); 2) analyse potentially toxic metabolites (blood, saliva, hair); 3) perform pharmacogenetic tests for non-functional polymorphisms of genes encoding key transport proteins and enzymes involved in drug metabolism. Results of pharmacogenetic and pharmacometabolic tests applied in the clinical practice of an epileptologist will allow to manage lamotrigine neurotoxiсity.
INTRODUCTION. Breast cancer (BC) ranks as second most lethal cancer type among female population. Triple-negative breast cancer (TNBC) is one of the most aggressive and treatment-resistant molecular biological subtypes. The metastatic form of triple-negative breast cancer (mTNBC) is an urgent problem in breast cancer therapy, since the treatment effectiveness depends on multiple factors. Analysis and assessment of adverse chemotherapy reactions is essential for high-quality medical care.
AIM. This study aimed to assess the risks of adverse drug reactions and the severity of their consequences during mTNBC chemotherapy in order to develop management and prevention strategies for these reactions.
MATERIALS AND METHODS. The methods included content analysis of scientific publications, regulations, Russian national standards of BC medical care in adults, clinical recommendations for adult BC therapy and a retrospective analysis of primary medical records covering BC patients at Astrakhan Regional Clinical Oncological Dispensary in 2023-2024.
RESULTS. The study established therapeutic strategies of the first- (doxorubicin + cyclophosphamide, docetaxel) and second-line drug therapy (paclitaxel + carboplatin, eribulin) for mTNBC. Established adverse drug reactions that occur after administration include febrile neutropenia, cardiotoxicity, peripheral neuropathy, drug resistance, nausea, and vomiting. The highest risk category for the two lines of therapy was assigned to febrile neutropenia, the lowest — to nausea and vomiting.
CONCLUSIONS. The incidence and profile of adverse reactions vary significantly depending on chemotherapy protocols and lines used in patients with mTNBC. Haematological toxicity being predominant for each of the protocols, anthracycline-cyclophosphamide strategy bears the highest risk of adverse drug reactions. Eribulin and docetaxel monotherapies are the safest options. The study results can serve as a basis for improving and optimising mTNBC chemotherapy.
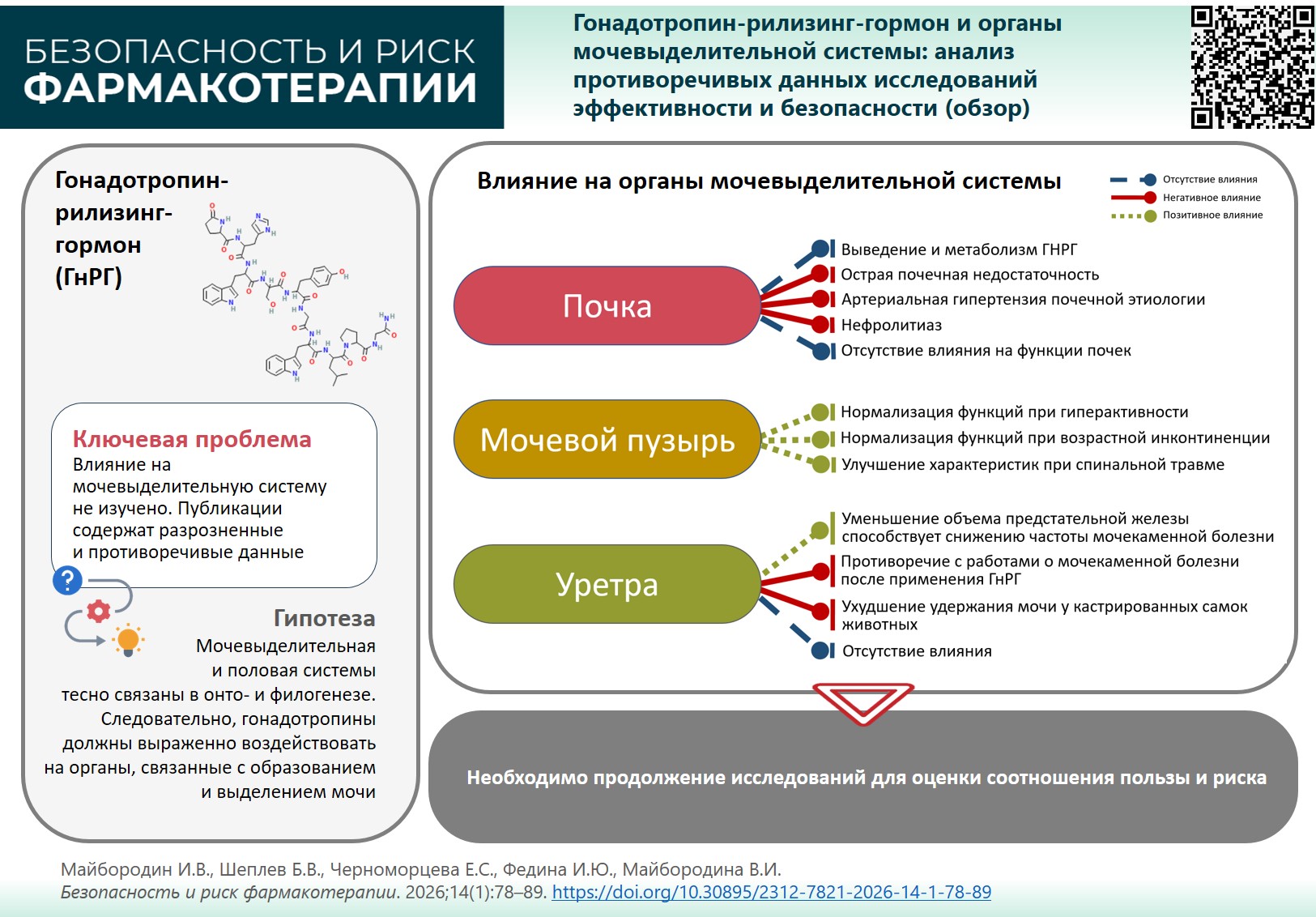
INTRODUCTION. Gonadotropin-releasing hormone (GnRH) products are used to treat reproductive system disorders both in females (for temporary medically induced menopause) and males (in order to suppress certain functions in benign and malignant diseases). The urinary and reproductive systems are closely linked in their ontogenesis and phylogenesis. A summary and critical analysis of the existing data will allow us to assess the effects of GnRH products on the urinary system and identify areas for further research.
AIM. This study aimed to identify benefits and risks of GnRH therapy for the urinary system in order to optimize hormone therapy in various conditions.
DISCUSSION. Analysed preclinical and clinical studies showed that GnRH and its metabolites are primarily excreted by the kidneys. GnRH therapy affects the renal system: nephrolithiasis may develop due to calcium olism disorders; cases of acute kidney injury, including acute renal failure, have been reported as well. Bladder structures include GnRH receptors, resulting in high GnRH binding to bladder cells and effective GnRH therapy. However, there are sex-specific differences; high GnRH and its receptors in bladder cancer are associated with better overall survival in men. High expression of GnRH receptors on cancer cells determines the success of GnRH therapy in the bladder tumours. GnRH analogues show a positive clinical effect in women with age-related menopausal incontinence, directly influencing bladder tissue, the sphincter system, and urethra. However, the data in some of the reviewed articles were insufficiently substantiated and lacked statistical validation. A number of studies reported no effect of GnRH therapy on renal and urethral function, or on bladder tumour outcomes.
CONCLUSIONS. The apparent lack of data and inconsistent publications on each aspect of GnRH influencing the urinary system indicate a paucity of research for this issue. Further applied and fundamental research of GnRH effects on the urinary tract is warranted to assess the benefit-risk ratio, develop less toxic anticancer agents, and effectively prevent and treat adverse reactions during hormone therapy for various diseases.
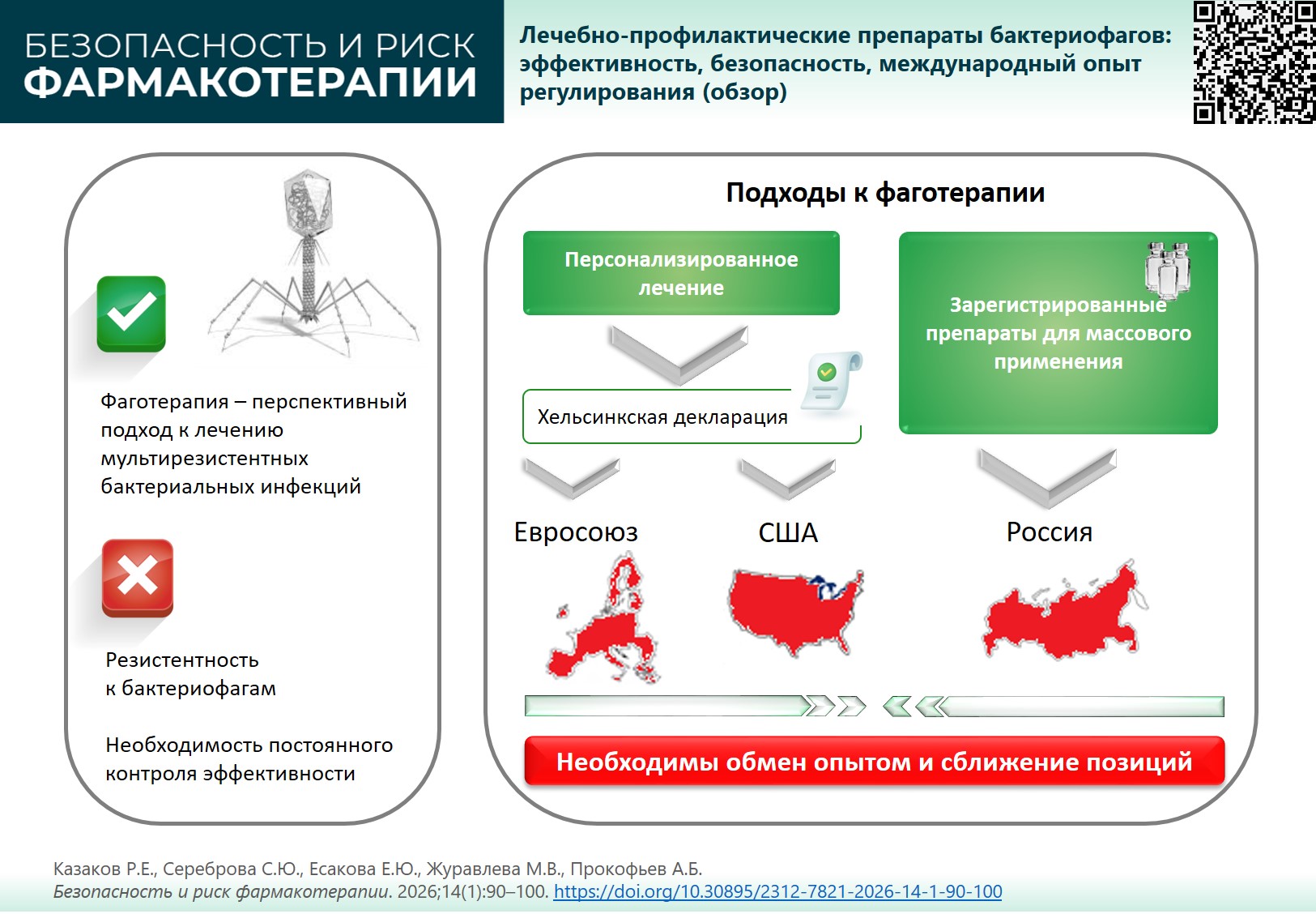
INTRODUCTION. Bacteriophage preparations are effective in bacterial infections, including multi-drug resistant pathogens. In order to develop and widely use standardized and personalized phage therapy, current regulations and guidelines governing the development, clinical trials, registration, and distribution of bacteriophage preparations necessitate an update.
AIM. This study aimed to analyze global trends in the development, registration, as well as efficacy and safety assessment of bacteriophage preparations to identify promising areas for development of phage therapy in the Russian Federation.
DISCUSSION. Despite the proven efficacy, rapidly expanding resistance of pathogens, complicated delivery to the infection focal point, and poorly understood safety of phage therapy prevent wide implementation of bacteriophages in clinical practice. In case standardized bacteriophage preparations show decreased effectiveness, a modified formulation may be warranted, with the updated strains; however, no appropriate legislative mechanisms exist so far. To date, 14 standardized bacteriophage preparations were registered and used in the Russian Federation, with completed clinical trials. Personalized phage therapy is used to a lesser extent. In the United States and the European Union (EU) member states, strict requirements for safety and efficacy evidence are placed; phage therapy is not an official treatment method but is used in individual cases. In some countries, particularly Poland and Belgium, phage therapy is under close supervision, while the use of bacteriophages remains individualized. In 2025, a draft of a concept document stipulating development and production of bacteriophage agents for human use was developed in the EU. However, none of the drugs for phage therapy was approved according to the EU and US requirements.
CONCLUSIONS. Russian Federation has a unique experience of using standardized phage preparations. Regulatory algorithms of modifying the formulation of a registered bacteriophage preparation are still to be developed both in the Russian Federation and other countries. Worldwide harmonization of the regulatory base is an essential element of global development; points to consider include new EU and US initiatives on developing regulatory documents, intensified research aimed at overcoming bacterial resistance to bacteriophages, and long-term safety studies.
INTRODUCTION. Lasmiditan, a new selective 5-HT1F receptor agonist (approved by FDA in 2019), relieves migraine attacks via neuronal inhibition and acts in the central and peripheral nervous systems. Unlike triptans (5-HT 1B/1D receptor agonists), the major group of preparations used in severe migraines, lasmiditan does not cause vasoconstriction.
AIM. This study aimed to evaluate the role of lasmiditan, a representative of a new class of neuroactive anti-migraine drugs (ditans), in migraine management based on its mechanism of action as well as effectivnes and safety review.
DISCUSSION. Anti-migraine mechanism of action of lasmiditan is due to its selective effect on 5-HT1F serotonin receptors at the trigeminovascular level and in central nervous system pain-modulating pathways.
Lasmiditan demonstrated efficacy superior to placebo in three randomized clinical trials (SAMURAI, SPARTAN, CENTURION, n=5,910). In the population of patients with high cardiovascular risk, pain relief was achieved in 31.4% patients (100 mg) and 38.8% (200 mg) compared to 21.3% in the placebo group; the most bothersome symptom was reversed in 44.2% (100 mg) and 48.7% (200 mg) compared to 33.5% in the placebo group. The sustained pain relief was maintained in 13.6% and 17.3% of patients (100 and 200 mg, respectively).
Adverse events had the central character and were dose-dependent: dizziness (14.7%), paresthesia (5.7%), somnolence (5.5%), fatigue (3.8%), nausea (3.4%), muscle weakness (1.3%), and hypoesthesia (1.2%); no patient groups (including coronary disease) showed cardiovascular complications. Most adverse events were mild to moderate, while their incidence decreased with the long-term use of lasmiditan. There were rare reports of serotonin syndrome in randomized clinical trials and post-marketing experience. Impaired alertness and response rate was also observed for 8 h after lasmiditan intake.
CONCLUSIONS. Lasmiditan may serve as an alternative to triptans in patients at high cardiovascular risk or with poor response to triptans. The potential for central nervous system adverse events should be assessed, alongside with the potential neuropsychiatric complications, concomitant therapy, and occupational factors.
INTRODUCTION. Chronic obstructive pulmonary disease (COPD) is a leading cause of death and significant economic losses for the healthcare system. While there is a trend towards decreasing COPD incidence in Europe, Russia saw the incidence increase by 5%, and the economic burden rose from 0.20% to 0.34% of GDP in 2022–2023. International and nationwide best practices necessitate a further analysis to identify the ways of reducing financial burden on the Russian healthcare system.
AIM. This study aimed to identify and evaluate feasibility for the most effective ways to reduce the financial burden when managing COPD patients in the Russian Federation.
DISCUSSION. Key differences in COPD epidemiology and cost structure were identified between Russia and EU countries. In the European Union (2001–2019), COPD incidence decreased among men (–9.7%) and increased among women (+4.3%) due to higher smoking rates. In Russia, the incidence continued to grow (+5%), while the mortality remained high (26% of the total death causes). In the EU, main direct costs were attributed to inpatient care (35–64%) and drug therapy (~25%), while the share of indirect costs reached 61–83%. Russian cost structure differed: the share of indirect costs was 92.6%, while among the direct costs, 76.1% was attributed to medicines supply, and only 18.7% covered inpatient care.
Effective burden reduction included: smoking cessation (reduced exacerbation risk by 39.7%; hazard ratio 0.65 for abstinence >10 years); prophylactic vaccinations (reduced hospitalizations by 50%, while vaccine refusal increased mortality 162-fold); modern pharmacotherapy (triple therapy: long-acting anticholinergic agents + long-acting beta2-agonists + inhaled glucocorticoids, reduced exacerbation frequency by 24% and mortality by 49%), and pulmonary rehabilitation (OR for hospital readmissions 0.44) still presented poorly in Russia (~10%).
CONCLUSIONS. To reduce COPD economic burden in the Russian Federation, it is necessary to increase the coverage with preventive measures (tobacco control programs, immunization), develop pulmonary rehabilitation programs using digital technologies, implement modern pharmacotherapy regimens, and improve physicians’ expertise via continuing medical education.
ISSN 2619-1164 (Online)