AUTHORITATIVE OPINION
Identification of the genetic features that determine a patient’s individual response to a medicinal product and provision of this information to a doctor helps to predict the effectiveness of therapy and the risk of adverse drug reactions. Dmitry A. Sychev — Academician of the Russian Academy of Sciences, Doctor of Medical Sciences, Full Professor, Rector of the Russian Medical Academy of Continuous Professional Education — talks about the importance of a personalised approach to selecting medicinal products and methods for their efficacy and safety control.
MAIN TOPIC: GENETICS AND PHARMACOLOGY: RESEARCH AND ADVANCES
INTRODUCTION. Pharmacogenetic (PGx) testing plays a significant role in predicting the risk of adverse drug reactions (ADRs) associated with valproic acid (VPA) products, which are among the most prescribed medicinal products in neurology and psychiatry. However, the sensitivity and specificity of PGx screening panels may be insufficient as individual valproate metabolism varies across ethnically/racially diverse patient populations.
AIM. The study aimed to identify implementation areas for a personalised approach to the development of PGx panels for assessing the safety and risk of valproate therapy in various ethnic and racial groups residing in the Russian Federation.
DISCUSSION. The authors reviewed the results of population studies concerning the frequency of non-functional and low-function alleles of genes encoding isoenzymes that play key roles in VPA P-oxidation in the liver. This review focused on studies published in eLIBRARY.RU, PubMed, Scopus, and Google Scholar in 2012–2022. The inclusion criteria were full-text original articles, systematic reviews, meta-analyses, Cochrane reviews, and clinical cases in Russian or English. The analysis revealed that the need for personalised assessment of the risk and safety of VPA may depend on the frequency of risk alleles for slowing down VPA P-oxidation in the liver across racial and ethnic groups worldwide, and particularly in Russia. The authors identified new areas to implement the personalised approach to the development of PGx panels for assessing the safety and risk of valproate therapy with consideration of the rates of hepatic VPA P-oxidation in patients of different ethnic and racial backgrounds. However, the review of population-based associative genetic research from around the world demonstrated the current lack of clarity in the prospects of translating international findings directly into Russian clinical practice through the development of PGx panels due to Russia’s ethnic/racial diversity and vast territory.
CONCLUSIONS. To increase the sensitivity and specificity of Russian PGx panels, bridging studies are required to extrapolate the associations established between the most common risk alleles and VPA P-oxidation disorders in other ethnic groups to a specific population of a specific Russian region.
INTRODUCTION. Drug-induced liver injury (DILI) is associated, among other things, with the use of antibiotics. Children with DILI are at risk of acute liver failure and even death. However, the literature on the subject provides little information on the possibility of distinguishing the types of hepatic lesions to diagnose potentially life-threatening DILI in time.
AIM. The study aimed to describe the phenotype of new-onset DILI associated with antibiotics in critically ill children with nosocomial infection.
MATERIALS AND METHODS. The authors conducted a prospective observational study in the resuscitation and intensive care units of the Morozov Children’s City Clinical Hospital from 1 February 2020 to 1 September 2021. The study assessed the incidence of antibiotic-associated DILI using the Global Trigger Tool. The study enrolled 100 critically ill children aged 0 to 17 years (44 boys and 56 girls) with nosocomial infection.
RESULTS. Signs of hepatotoxicity were detected in 19 patients, including 8 with abnormal liver function tests but normal liver function and 11 with abnormal liver function tests and clinically apparent liver disease. Thus, the incidence of new-onset hepatotoxicity associated with antibiotics amounted to 12.9 cases per 100,000 paediatric patients, and the incidence of DILI was 7.5 cases per 100,000 children. Based on the analysis of medical records, biochemical findings, and relationships between the time of dosing and the manifestation of signs of liver disorder in 11 children, the authors characterised the phenotype of idiosyncratic cholestatic hepatitis. Critically ill children treated with antibiotics showed alanine transaminase activity up to 10 times the upper limit of normal (ULN), bilirubin levels up to 4.45 times the ULN, and gamma-glutamyl transferase activity up to 5 times the ULN. The odds of developing new-onset DILI were the highest with tigecycline (OR: 4.07; 95% CI: 1.32–12.50) and meropenem (OR: 1.84; 95% CI: 1.01–3.36). In 6 patients, clinical signs of idiosyncratic cholestatic hepatitis resolved within a few weeks after antibiotic discontinuation. The other 5 patients with clinical signs of idiosyncratic cholestatic hepatitis died.
CONCLUSIONS. The authors described the phenotype of idiosyncratic cholestatic liver injury associated with antibiotics in critically ill children. The role of pharmacogenetic markers in the development of DILI associated with antibiotics in critically ill children needs to be assessed further to implement a risk-based approach and mitigate the risks.
The study was registered at ClinicalTrials.gov under No. NCT04141657 on 24 October 2019.
INTRODUCTION. The pathogenesis of antibiotic-associated drug-induced liver injury (DILI) in children has not been fully elucidated to date. Certain genotypes in patients increase the probability of developing DILI. Therefore, the identification of pharmacogenetic markers associated with DILI in children is essential.
АIM. This study aimed to identify pharmacogenetic biomarkers of new-onset DILI associated with tigecycline and meropenem in children.
MATERIALS AND METHODS. This prospective observational study was conducted in the Morozov Children’s City Clinical Hospital from 1 February 2020 to 1 September 2021. The study analysed the incidence and types of antibiotic-associated adverse drug reactions (ADRs) in 100 critically ill children aged 0 to 17 years (44 boys and 56 girls). Pharmacogenetic testing was performed in children with ADRs (n=30) to identify potential mechanisms involved in the development of their ADRs. The authors isolated and tested DNA from buccal epithelium swabs using the Agena Bioscience iPLEX® PGx Pro-based VeriDose® Core Panel covering 68 single nucleotide polymorphisms (SNPs) or short insertions and deletions (INDELs) and 5 copy number variants (CNVs).
RESULTS. The odds of developing DILI associated with meropenem and tigecycline were higher in carriers of the homozygous cytochrome genotype CYP3A5*3/*3 (OR: 12.6; 95% CI: 1.9–79.4, r=6.54, p=0.011) than in patients with the heterozygous genotype CYP3A5*1A/*3. The odds were even higher in patients not carrying the CYP3A5*1A/*3 genotype (OR: 17.14; 95% CI: 1.79–16.3, r=6.24, p=0.013). The detection of the CYP3A5*3/*3 genotype had a prognostic accuracy of 76.7%, a sensitivity of 82%, and a specificity of 74% in predicting the risk of DILI associated with meropenem and tigecycline. Moreover, children with DILI carried the heterozygous genotype SLCO1B1*1/*5 (rs4149056 polymorphism) more often than children with other adverse reactions associated with meropenem and tigecycline (r=9.8, p=0.002).
CONCLUSION. The results of this study prove the prognostic significance of the homozygous cytochrome genotype CYP3A*3/*3 as an indicator of a potential risk for developing DILI associated with meropenem and tigecycline in children in critical conditions.
The study was registered at ClinicalTrials.gov under No. NCT04141657 on 24 October 2019.
INTRODUCTION. The variability in patient response to non-steroidal anti-inflammatory drugs (NSAIDs) may be due to genetic polymorphisms. Decreased-function or non-functional CYP2C9 alleles are known to be significant contributors to response variability, and research is being conducted on the potential contribution of polymorphisms in the PTGS1 and PTGS2 genes that encode for the NSAID targets, cyclooxygenases 1 and 2.
AIM. This study aimed to demonstrate the contribution of pharmacogenetic characteristics to the development of adverse reactions to NSAIDs in clinical cases.
MATERIALS AND METHODS. This study reports two clinical cases observed in a multidisciplinary hospital in Moscow. The first case report demonstrates a neurosurgical patient with spondylodiscitis who showed signs of renal failure associated with ketorolac administration during 13 days of inpatient treatment. The second case report presents a patient with gastrointestinal bleeding that developed after 3 weeks of ketorolac self-administration and ultimately required gastric resection surgery. According to real-time polymerase chain reaction results, these patients have single-nucleotide polymorphisms, including rs179985 (CYP2C9*2) and rs1057910 (CYP2C9*3) in the CYP2C9 gene, rs10306135 and rs12353214 in the PTGS1 gene, and rs20417 in the PTGS2 gene.
RESULTS. The described adverse reactions to ketorolac are attributable to the patients’ pharmacogenetic characteristics and other factors (exceeding the recommended duration of ketorolac therapy, concomitant conditions, drug interactions, and older age). In the case of nephrotoxicity, the patient has the CYP2C9*1/*2 genotype, which is associated with decreased NSAID metabolism. Additionally, this patient has the PTGS1 (C>T) rs12353214 genotype, which may be associated with variations in the activity of cyclooxygenase 1 and may lead to an increased risk of adverse reactions to NSAIDs. In the case of gastrointestinal bleeding, the patient also has genetic characteristics that affect the response to NSAIDs, as the CYP2C9*1/*3 genotype is associated with significantly decreased NSAID metabolism. Patients with this genotype should take the lowest doses or switch to alternatively metabolised NSAIDs.
CONCLUSIONS. To reduce the risk of nephrotoxicity and gastrointestinal bleeding in patients when prescribing ketorolac, healthcare providers should strictly follow the recommended dosing regimen and consider individual comorbidities and potential drug interactions that may potentiate adverse drug reactions. Additionally, patient risk management requires pharmacogenetic testing, including the determination of decreased-function or non-functional CYP2C9 alleles and the rs12353214 polymorphism in the PTGS1 gene.
RATIONAL PHARMACOTHERAPY AND PHARMACOVIGILANCE
INTRODUCTION. Osteoporosis is a leading cause of morbidity, disability, reduced quality of life, and premature mortality in the elderly population. Denosumab is a treatment for osteoporosis; however, denosumab discontinuation may cause a rebound effect, which is a severe adverse drug reaction (ADR) leading to an increase in the rate of bone tissue loss. Studying the mechanism of the rebound effect and potential ways to manage it can improve the safety of denosumab therapy.
AIM. This study aimed to summarise up-to-date information regarding the mechanism of the rebound effect and ways to manage it after denosumab discontinuation.
DISCUSSION. Osteoporosis results from an imbalance in bone remodelling. Recent studies have shown that mature osteoclasts are able to fission into osteomorphs, which separate from the polykaryon and fuse with neighbouring osteoclasts or other osteomorphs (osteoclast recycling). The mechanism of action of denosumab is based on the inhibition of osteoclast recycling, which results in the accumulation of pre-osteoclasts and osteomorphs. Research into processes occurring in bone tissue shows that denosumab creates a pool of induced cells, and when denosumab therapy is discontinued, these induced cells quite quickly and abundantly differentiate into osteoclasts causing bone resorption (rebound effect) and increasing the risk of fractures. In order to improve mineral bone density and to prevent fractures after denosumab discontinuation, it is reasonable to use antiresorptive medicines from the bisphosphonate class. Bisphosphonates accumulate in bone tissue and concentrate in areas of active bone metabolism.
CONCLUSIONS. Further studies of the rebound effect mechanism, including a deeper understanding of the role of osteomorphs in osteogenesis, will improve the measures taken to reduce the risk of fractures after denosumab discontinuation. Further research is needed to evaluate the effect of antiresorptives on bone tissue loss after denosumab discontinuation.
INTRODUCTION. A risk management plan (RMP) is an important document for assessing and managing risks when using a medicinal product. An RMP is provided as part of the marketing authorisation application for a medicinal product. The requirements for the need to submit an RMP and the information in its sections depend on the type of medicinal product and marketing authorisation procedure. Marketing authorisation holders will benefit from an expert systematisation of the requirements for RMP submission outlined in the Eurasian Economic Union (EAEU) Rules for Marketing Authorisation and Expert Assessment of Medicinal Products for Human Use and Rules of Good Pharmacovigilance Practice (Eurasian Economic Commission Council Decisions Nos. 78 and 87 dated 03.11.2016, respectively), as this systematisation will contribute to a more accurate drafting of RMPs.
AIM. This study aimed to analyse the requirements for submitting parts and modules of an RMP depending on the type of medicinal product being authorised according to the EAEU marketing authorisation procedures.
RESULTS. This review analyses and systematises the requirements for the development of an RMP and the submission/non-submission of its main parts (I–VI) and Part II modules (CI–CVIII) depending on the type of medicinal product and marketing authorisation procedure. The article describes the principles for providing information in three main RMP sections: Safety Specifications (Part II), Pharmacovigilance Plan (Part III), and Risk Minimisation Measures (Part V). Flow charts illustrate summarised information on pharmacovigilance activities and risk minimisation measures that is included in an RMP. The article systematises the considerations for the analysis and evaluation of information for the main parts (I–VI) and Part II modules (CI–CVIII) of an RMP.
CONCLUSIONS. The systematisation of the requirements of Eurasian Economic Commission Council Decisions Nos. 78 and 87 dated 03.11.2016, as well as the expert comments on considerations for RMP submission depending on the type of medicinal product and marketing authorisation procedure will help to improve the quality of RMPs and, in general, the safety of medicinal products authorised in the EAEU.
INTRODUCTION. Polycystic ovary syndrome (PCOS) is the main cause of female infertility. Clinical practice guidelines on PCOS recommend combined oral contraceptives (COCs) as first-line therapy. Given the association of PCOS with a number of clinical conditions, including obesity, type 2 diabetes mellitus, metabolic syndrome, anxiety and depressive disorders, an increased risk of endometrial cancer, and cardiovascular diseases, it is important to describe and systematise data on the benefits and risks associated with COCs.
AIM. This study aimed to review current data on the use of COCs in PCOS, focusing on the rationality of prescribing, the main risks of using COCs, and the measures to minimise these risks.
DISCUSSION. Hirsutism is the most prominent clinical manifestation of hyperandrogenism; in PCOS, its prevalence is 65–75%. COCs containing progestins with anti-androgenic properties are effective in treating hirsutism and superior to anti-androgens alone. Women with PCOS are at increased risk of endometrial cancer due to prolongation of the oestrogen-dependent proliferative phase. This risk can be mitigated by the normalising effect of COCs on the menstrual cycle and the effect of the progestin component of COCs. This review highlights the controversial nature of data on the association of COCs with emerging or exacerbating mental disorders, impaired glucose tolerance, and arterial hypertension/other cardiovascular risks in PCOS. Since the main safety issue with COCs is their thrombogenicity, clinicians should take special care when prescribing medicinal products from this group to PCOS patients. Data on the effect of COCs on fertility in women with PCOS are limited, but there is published evidence of an adverse effect of long-term use of COCs on the results of assisted reproductive technologies.
CONCLUSIONS. The key benefit of the non-contraceptive use of COCs in PCOS is in treating hyperandrogenism, mainly hirsutism. To achieve effect, the patient should take COCs containing progestins with anti-androgenic properties for at least 6 months. PCOS patients can also use COCs for contraception. Clinicians prescribing COCs to women with PCOS should consider the individual patient context, the aim of treatment, and any potential risks associated with COCs.
QUALITY OF MEDICINES
INTRODUCTION. Gelatine capsules are an essential dosage form used for many medicinal products. Gelatine capsules are subject to quality control in accordance with the requirements of the general monograph on capsules of the State Pharmacopoeia of the Russian Federation. This monograph does not standardise the content of elemental impurities, as these impurities are controlled at the raw material level. However, the equipment and excipients used in the production of gelatine capsule shells may be additional sources of impurities. Preliminary screening for chemical elements will help to assess the need for limiting elemental impurities in gelatine capsule shells.
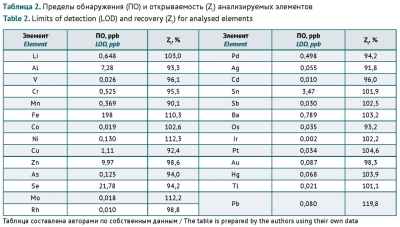
AIM. This study aimed to identify the chemical elements that contribute to gelatine capsule shell contamination the most by inductively coupled plasma mass spectrometry, to list the elements requiring specification, and to limit their content from a risk-based pharmaceutical quality control perspective.
MATERIALS AND METHODS. The study used 18 samples of hard gelatine capsules by different national and international manufacturers. The elemental analysis was performed using an Agilent 7900 inductively coupled plasma mass spectrometer (Agilent Technologies).
RESULTS. The As, Ba, Co, Cr, Cu, Li, Mo, Ni, Pb, Sb, and V content in gelatine capsule shells did not exceed the permitted daily exposure (PDE) thresholds for oral dosage forms specified in the State Pharmacopoeia of the Russian Federation. Ag, Au, Cd, Hg, Ir, Os, Pd, Pt, Rh, Se, Sn, or Tl were not detected in any of the samples within the established detection limits. The Fe content in 67% of the tested shell samples was above the safety-based PDE for medical gelatine.
CONCLUSIONS. The greatest contributors to gelatine capsule shell contamination are Al, Fe, and Zn. The lack of established PDE thresholds for these elements presents a challenge in assessing the risks of adverse effects associated with ingesting these elements with gelatine capsules.
ISSN 2619-1164 (Online)